 
A cell placed into a hypotonic solution will swell and expand until it eventually burst through a process known as cytolysis. In hypotonic solutions, there is a net movement of water from the solution into the body. A hypotonic tonic solution is any external solution that has a low solute concentration and high water concentration compared to body fluids. 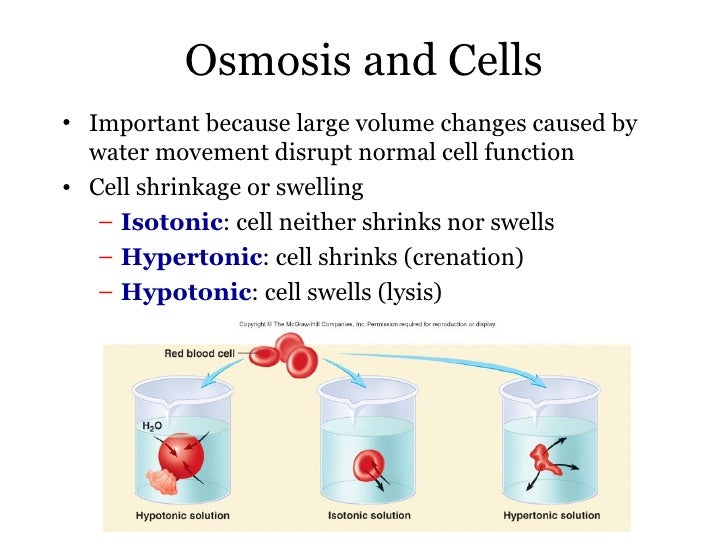
In an isotonic solution, no net movement of water will take place. An isotonic solution is any external solution that has the same solute concentration and water concentration compared to body fluids. A cell placed into a hypertonic solution will shrivel and die by a process known as plasmolysis. In a hypertonic solution, the net movement of water will be out of the body and into the solution. A hypertonic solution is any external solution that has a high solute concentration and low water concentration compared to body fluids. The process is illustrated by comparing an environmental or external solution to the internal concentration found in the body. The rate of osmosis always depends on the concentration of solute. One can think of osmosis as a specific type of diffusion. Both osmosis and diffusion are passive processes and involve the movement of particles from an area of high to low concentration. Diffusion is the net movement of particles from an area of high to low concentration. The concept of osmosis should not be confused with diffusion. Reverse osmosis is commonly used to purify drinking water and requires the input of energy. This flow is only possible with the application of an external force to the system. In reverse osmosis, water flows into the compartment with lower osmotic pressure and higher water concentration. Reverse osmosis occurs when water is forced to flow in the opposite direction. There is also thought that the interaction of solute particles with membrane pores is involved in generating a negative pressure, which is the osmotic pressure driving the flow of water. The compartment with the greatest osmotic pressure will pull water in and tend to equalize the solute concentration difference between the compartments. The physical driving force of osmosis is the increase in entropy generated by the movement of free water molecules. A reflection coefficient of 0 means a solute can freely permeable, and the solute can no generate osmotic pressure across the membrane. 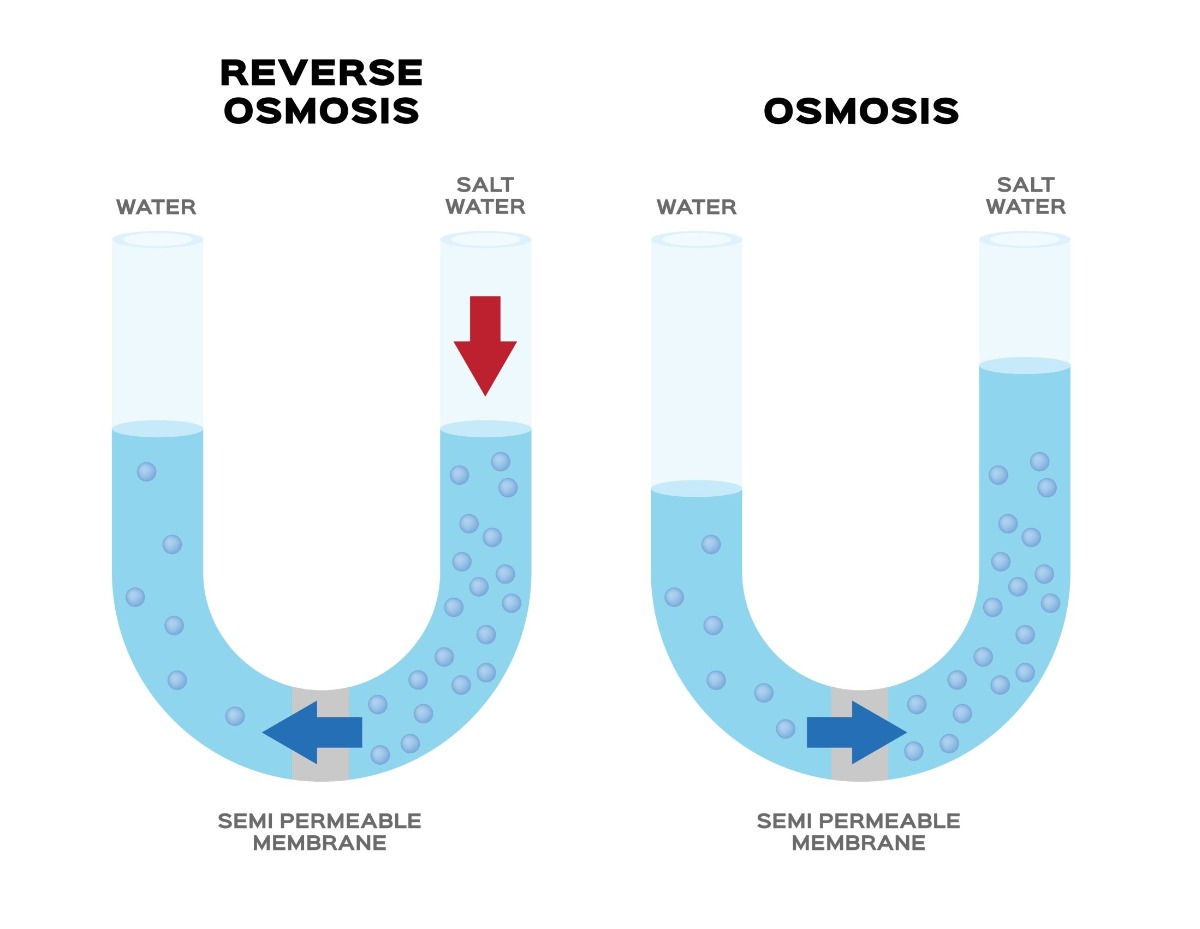
A reflection coefficient of 1 means a solute is impermeable. The reflection coefficient of a semipermeable membrane describes how well solutes permeate the membrane. 
The osmolality of a solution describes how many particles are dissolved in the solution. Its measured osmolality can describe the osmotic pressure of a solution. It is also true that, at a specific moment in time, water molecules can move towards either the higher or lower concentration solutions, but the net movement of water will be towards the higher solute concentration. The compartment with the highest solute and lowest water concentration has the greatest osmotic pressure. Osmotic pressure can be calculated with the van 't Hoff equation, which states that osmotic pressure depends on the number of solute particles, temperature, and how well a solute particle can move across a membrane. Osmosis can still occur with some permeability of solute particles, but the osmotic effect becomes reduced with greater solute permeability across the semipermeable membrane. It is important to emphasize that ideal osmosis requires only the movement of pure water across the membrane without any movement of solute particles across the semipermeable membrane. Across this membrane, water will tend to move from an area of high concentration to an area of low concentration. In physiology, osmosis (Greek for push) is the net movement of water across a semipermeable membrane. 
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
